An amu is equivalent to one dalton unit. Because the molecular mass of Hydrogen is 1grammole there is 1 mole of hydrogen in 1 gram of hydrogen atoms.

Average Atomic Mass Video Khan Academy
In kilograms the molar mass of a hydrogen atom can be written as 0001 kilograms per mole.
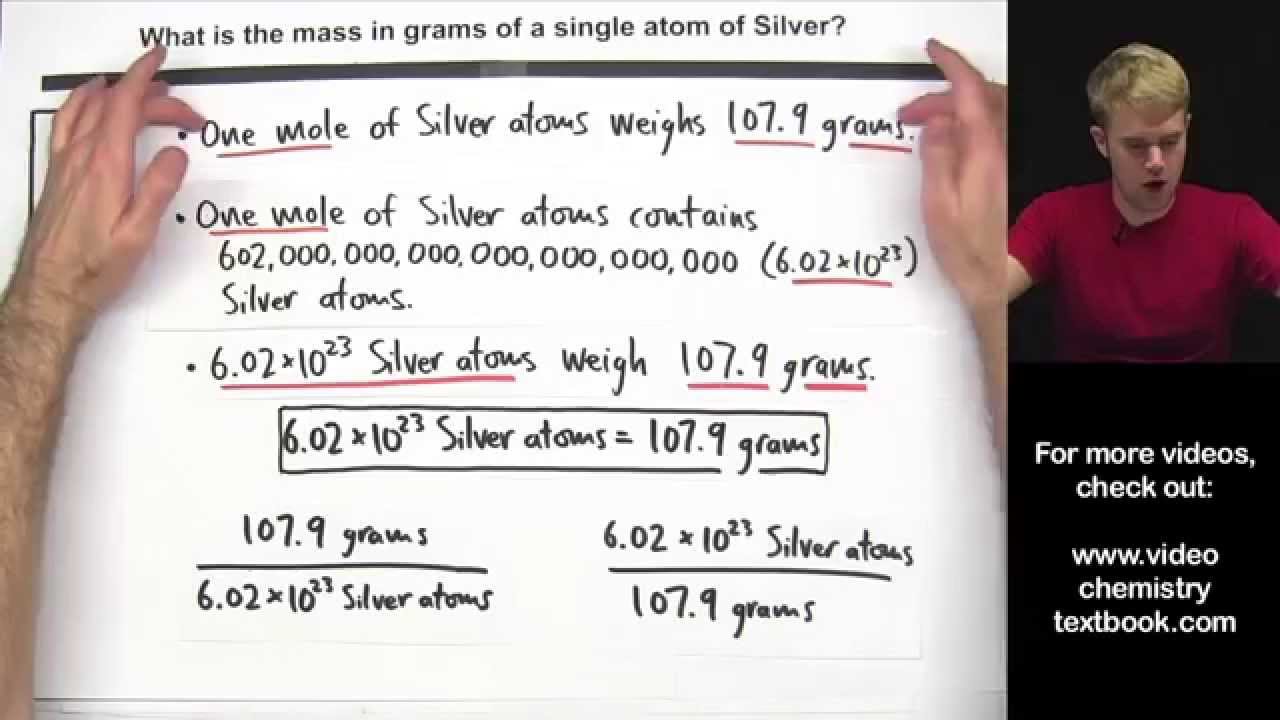
What is the mass of one hydrogen atom in grams. The weight of 1 atom of hydrogen is calculated as 166058 10-24 grams. The atomic mass of an element expressed in grams is called gram atom. Thus weight of 1 atom of hydrogen is 60210 231008.
A relative scale can be devised by assuming that one of the elements has exactly one mass unit for each of its nucleons. We have to find the mass of 1841025 184 10 25 atoms of. Problem 120 Easy Difficulty.
Molar mass can also be defined as the molecular mass of the given substance expressed in grams. So then why isnt the atomic mass of Hydrogen exactly 1. The number of grams given on the periodic table represents the mass of one mole of that element.
It completely depends on what element. The atomic mass is useful in chemistry when it is paired with the mole concept. What is the empirical formula of this compound.
X 166058 10 -24. If the mass of one mole of hydrogen is 101 grams. 1 atom of 12 C has a mass of 12 amu 1 mol 12 C has a mass of 12 g.
The mass of one hydrogen atom is 167 times 10-24 gram. Some Notes about Gram Atomic Mass. For every mole there are 6021023 atoms so in 1 gram of hydrogen there are 6021023 hydrogen atoms in scientific notation this is equal to 602000000000000000000000 hydrogen atoms.
Weight of 6022 10 23 atoms of hydrogen 1008 g. Whereas one hydrogen atom has a mass of approximately 1 u 1 mol of H atoms has a mass of approximately 1 gram. Find the mass of 80000 hydrogen atoms.
What is the mass in grams of 111022 atom of gold Au. What is the mass of a single atom The mass of a single atom of an element amu is numerically equal to the mass g of 1 mol of that element regardless of the element. Express the answer in scientific notation.
Hence the mass of 1 carbon atom is 19933 times text1text0 - 23gram Note- Molar mass is the mass of Avogadros number of atoms of that element or compound. A C 2 H 6 O B C 3 H 5. See full answer below.
For example the mass of one atom of hydrogen is about 1004 gramsmole but the mass of one atom of Uranium is. For example when I look at hydrogen on the periodic table the mass is 10079. 1 gram atom of hydrogen 1008 g 1 mole of hygrogen atoms.
The mass of a hydrogen atom is 16731024 g 1673 10 24 g. Thus since the atomic mass of iron is 55847 amu one mole of iron atoms would weigh 55847 grams. The atomic mass of an element measured in amu is the same as the mass in grams of one mole of an element.
We round to two figures on this website. X 1 6022 10 23. If you check a periodic table youll see that Hydrogen actually has a mass of 100794.
As 6022 10 23 atoms of hydrogen weighs 1008g 1 atom of hydrogen weighs. When calculating the gram atomic mass remember to round to the appropriate number of figures. Avogadros number is 6022 times 1023 and can be used to represent the number of representative particles of any particle on the atomic scale.
And whereas one sodium atom has an approximate mass of 23 u 1 mol of Na atoms has an approximate mass of 23 grams. The mass of an atom of hydrogen can also be expressed in molar mass units as one gram per mole. A 22 B 28 C 36 D 39 Solution- Atomic weight for.
Three mols of hydrogen atoms 15 mol of hydrogen gas has a mass of 303 g. For every mole there are 6021023 atoms so in 1 gram of hydrogen there are 6021023 hydrogen atoms in scientific notation this is equal to 602000000000000000000000 hydrogen atoms. In grams the mass of an atom of hydrogen is expressed as 167 x 10 -24.
We now know that a hydrogen atom has a mass of 16735 x 10-24 grams and that the oxygen atom has a mass of 26561 X 10-23 grams. The mass of 1 mole of C-12 atoms 12 g 1 mole of C-12 atoms 6022 10 23 atoms The mass of 1 atom of C-12 12 6022 10 23 199 10-23 g. The atomic mass of hydrogen is 101.
Watch the video for more information on size of an atom. Is the molecular mass of hydrogen gas therefore as per the definition the molar mass of hydrogen gas will be 2 g. And oxygen 160 grams.
60210 23atoms of hydrogen weigh 1008 g. If hydrogen is the lightest of all substances then why not give it a mass of exactly 1 on our relative mass scale. It means that 1 gram atom of different elements has different masses.
Because the molecular mass of Hydrogen is 1grammole there is 1 mole of hydrogen in 1 gram of hydrogen atoms. It is also called a mole. 1 gram atom of carbon 120 g 1 mole of carbon.
Only carbon hydrogen and oxygen produced 2265 g of CO 2 and 1236 g of H 2 O. What is the mass of one atom of hydrogen.
How Many Times Is One Hydrogen Atom Heavier Than An Electron Quora
How Come The Mass Of A Single Atom Of An Element In Amu Is Numerically Equal To The Mass Of One Mole Of Those Atoms In Grams Quora
